Our Research
The Porosoff group develops new catalysts and methodologies for upgrading C1 and C2 resources into value-added chemicals, plastics, and fuels. We combine controlled synthesis, in situ spectroscopy, pulsed Joule heating, and AI-driven discovery to understand and engineer catalyst structure–activity relationships at a fundamental level.
Experimental work draws on catalyst synthesis and characterization, reactor studies, in situ X-ray absorption fine structure (XAFS) spectroscopy, FTIR, luminescence thermometry, and Joule reactors — combined with computational tools and machine learning.
CO₂ Hydrogenation to Plastics, Chemicals & Fuels
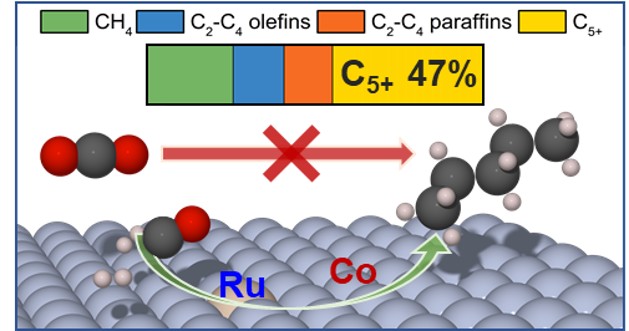
Through catalytic reactions, waste CO₂ can be converted to valuable oxygenates, specialty chemicals, and hydrocarbons for synthetic fuel. The current industrial approach uses two reactors in series: high-temperature reverse water-gas shift (RWGS, CO₂ + H₂ ↔ CO + H₂O), followed by Fischer-Tropsch synthesis (nCO + 2nH₂ → CₙH₂ₙ + nH₂O). The Porosoff group investigates novel dual-functional catalysts to selectively produce a tight distribution of hydrocarbons from CO₂ in a single step.
Recent work has established tungsten carbide (WC) as a viable earth-abundant alternative to platinum-group catalysts. By controlling the carburization kinetics and maintaining particle sizes below 10 nm, we demonstrated that the β-W₂C phase — selectively synthesized via temperature-programmed carburization — is an active and stable RWGS catalyst. This phase control framework resolves longstanding ambiguities in WC structure–property relationships and opens new pathways for low-cost CO₂ conversion.
We also investigate dual functional materials (DFMs) that combine CO₂ adsorption and hydrogenation into a single material, enabling integrated carbon capture and utilization (ICCU) with substantially reduced infrastructure.
Pulsed Joule Heating for Catalyst Synthesis & Electrification
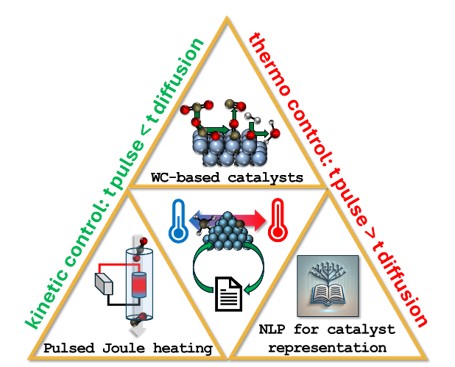
Transforming the chemical industry to meet US energy independence goals requires electrification of high-temperature catalytic processes for compatibility with intermittent renewable energy sources. Our group exploits the ultra-rapid heating and cooling rates of pulsed Joule reactors — capable of reaching extreme temperatures on millisecond timescales — to access catalyst phases that are kinetically trapped and otherwise inaccessible through conventional furnace-based synthesis.
For tungsten carbide, we hypothesize that Joule heating enables synthesis of entropically favorable WC phases by exploiting rapid heating rates to control carburization timescales. The hypothesized active phase for RWGS, orthorhombic WC (β-W₂C), is less thermodynamically stable than hexagonal WC (δ-WC), but possesses electronic features similar to the highly active β-Mo₂C — making WC an ideal model system for our study.
Beyond synthesis, we leverage Joule heating for transient catalysis experiments, using temperature oscillations to probe RWGS kinetics and control mass transfer, surface coverages, and catalyst restructuring in ways not accessible with conventional reactors.
AI & Natural Language Processing for Catalyst Discovery
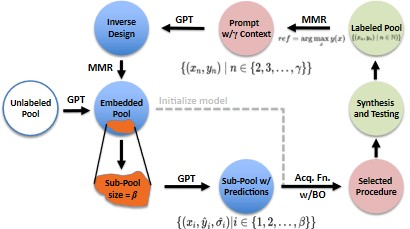
AI-directed design of experiments is transforming catalysis. Instead of relying on traditional structural and electronic features requiring expensive, difficult characterization, we leverage large language models (LLMs) to represent catalysts by the text of their synthesis procedures and reaction conditions. This approach is recently made possible by advances in LLMs and naturally captures physical-chemical details — including those due to catalyst restructuring during reaction — through the experimental conditions embedded in the text.
Combined with Bayesian optimization (BO), this NLP-based workflow accelerates discovery of earth-abundant, active, and selective catalysts for RWGS and beyond, with a current focus on trimetallic systems that are particularly difficult to characterize structurally. Trimetallic catalysts are an ideal fit for a text-based approach precisely because their complexity makes conventional descriptor-based modeling intractable.
The project also assesses the effects of experimental artifacts and irreproducible results on model performance, and works to establish standards for rigorous data reporting in catalysis. The broader methodology can be adapted to any process expressible in language — extending well beyond WC-based catalysts. Our recent work in ACS Central Science (2026) demonstrates Bayesian optimization of catalysis with in-context learning, using LLMs to propose experiments directly from natural-language descriptions of prior results.
Thermal Coupling & Localized Temperature Measurement in Catalytic Systems
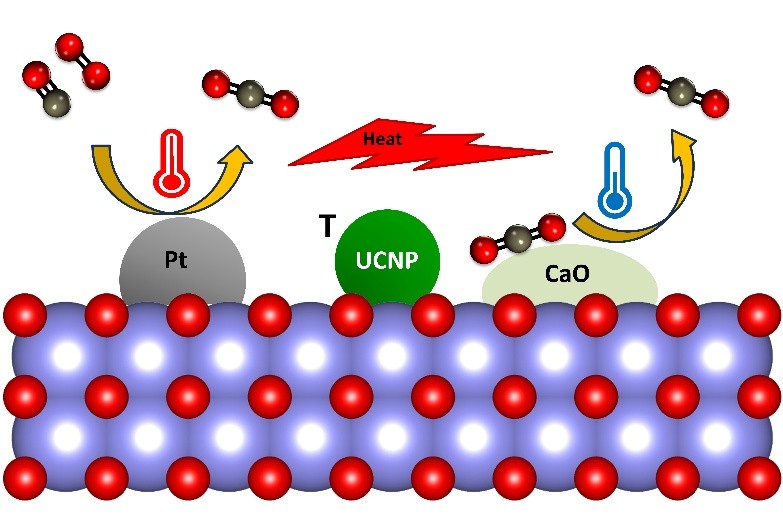
Chemical reactions are inherently thermal — some release heat (exothermic), others require it (endothermic). In tandem catalytic systems, exothermic and endothermic reactions can be strategically coupled so that heat released by one step drives another. However, precisely managing this thermal coupling requires knowing the actual temperature at the catalyst surface — not just in the bulk reactor bed.
In collaboration with the Pickel group (Department of Mechanical Engineering, University of Rochester), we have pioneered the use of upconverting nanoparticle (UCNP)-based luminescence thermometry to directly measure catalyst surface temperatures under in situ reaction conditions. This technique revealed that bulk temperature readings can differ from true surface temperatures by 10–100°C — a finding with major implications for measurement reproducibility, catalyst design, and cross-lab comparability in the field.
These thermal mapping tools enable us to identify and leverage hot spots on catalyst surfaces, optimize the spatial and temporal coupling of tandem reactions, and develop more energy-efficient catalytic processes. This methodology is broadly applicable to any thermally coupled system and is expected to improve reproducibility across the catalysis community.